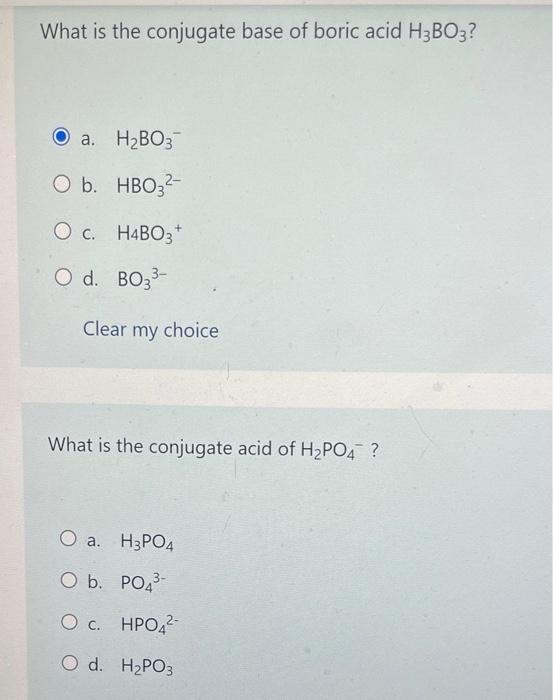
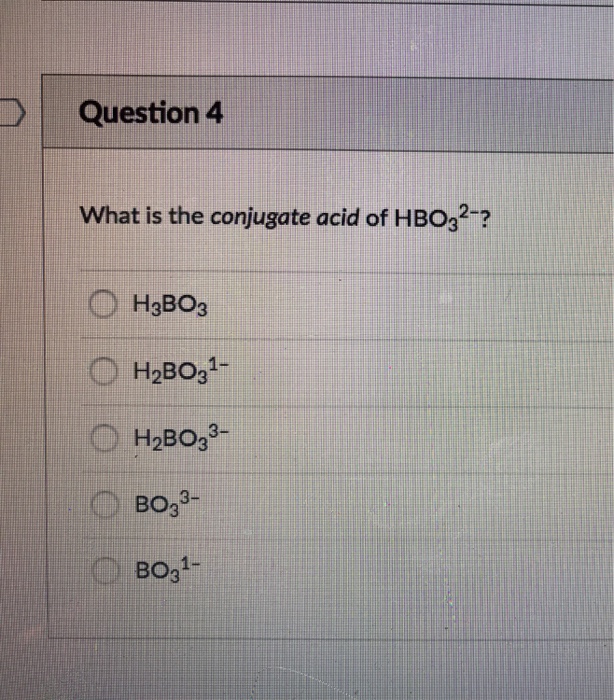
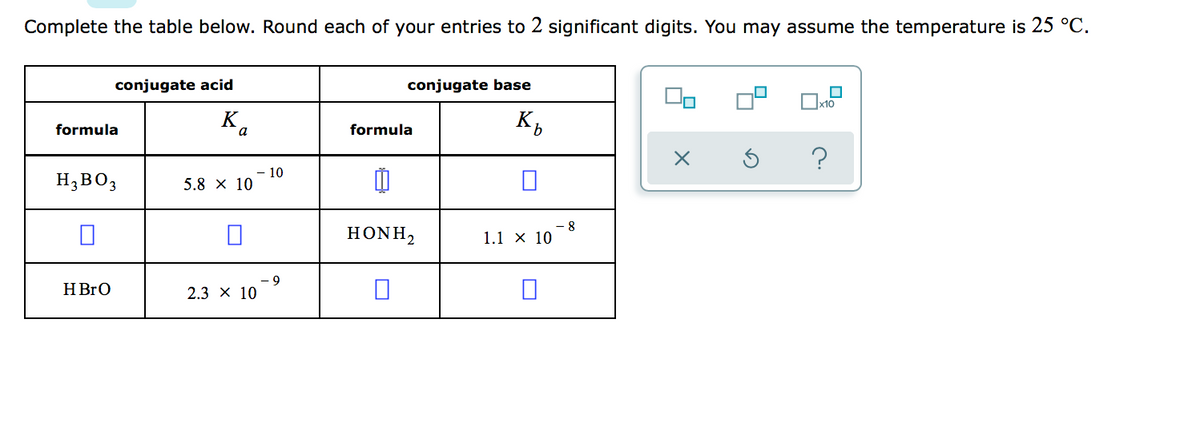
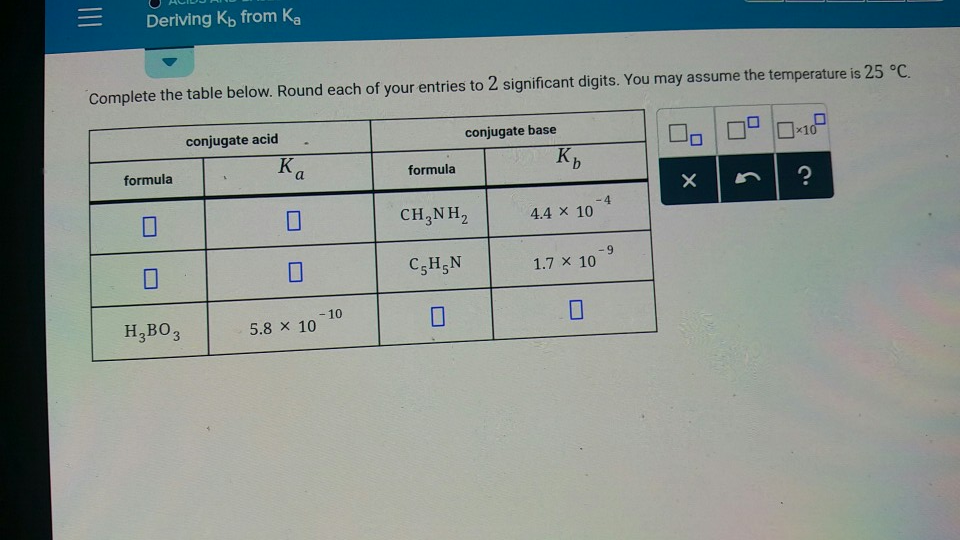
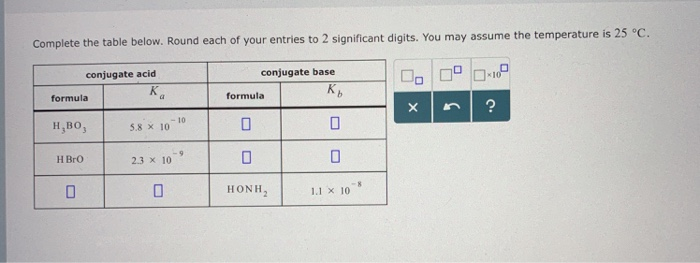
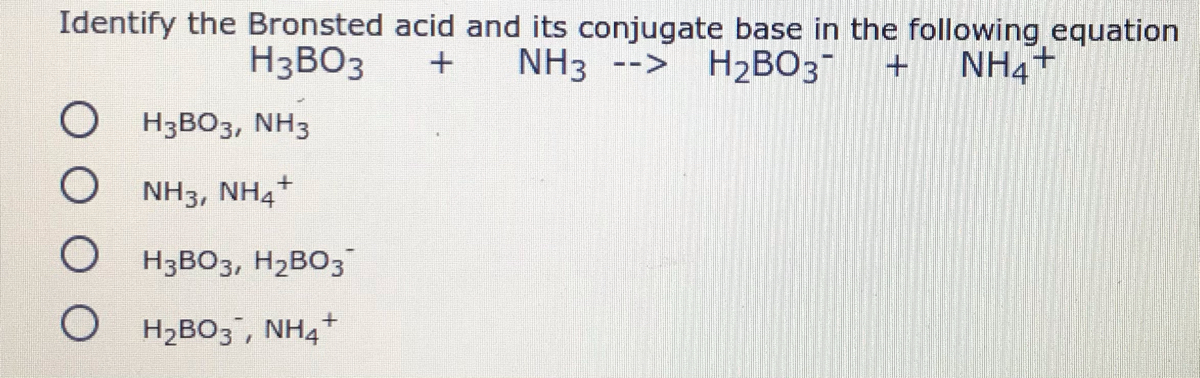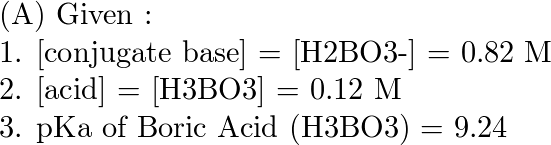Doubt: in this conjugate base is asked so H3BO3 will donate one proton so i think it should be H2BO3- Chapter: Equilibrium - Subject: Chemistry - Course: NEET Course - Complete Syllabus

SOLVED: 2. (a) For the following reactions, identify the acid, base, conjugate acid and conjugate base. (i) CH2ClCOOH + CH3COOH ⇌ CH2ClCOO‑ + CH3COOH2 (ii) H2SO4 + HNO3 ⇌ HSO4- + H2NO3+ (