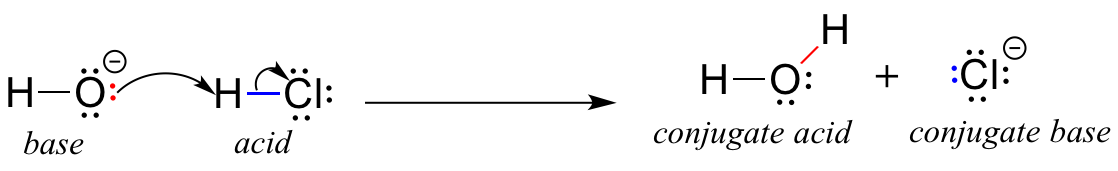
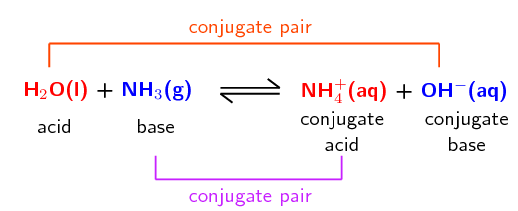
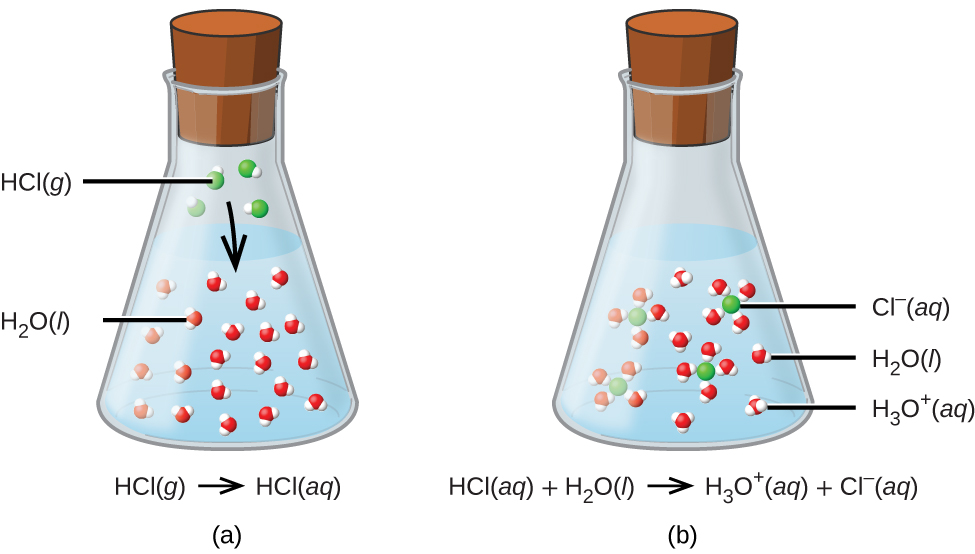
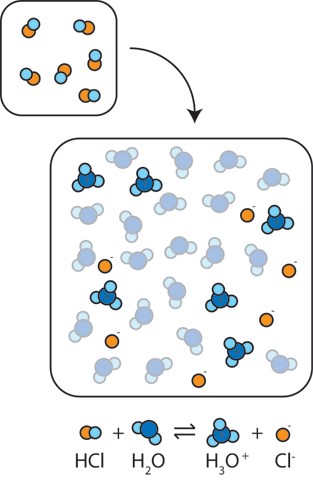
Anatomy and Physiology, Energy, Maintenance, and Environmental Exchange, Fluid, Electrolyte, and Acid-Base Balance | OERTX
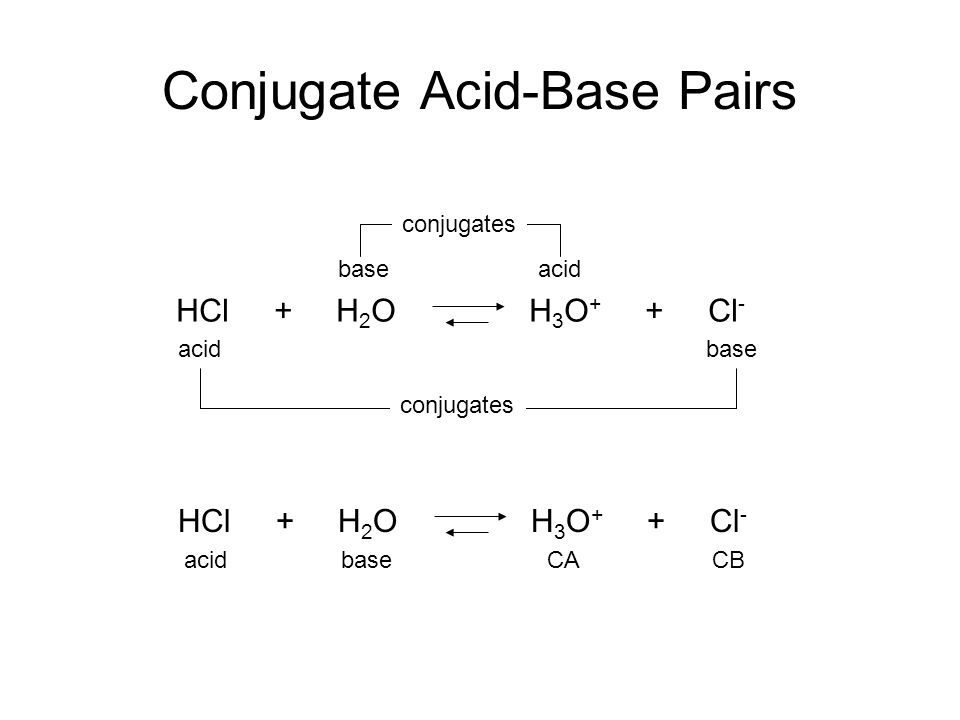
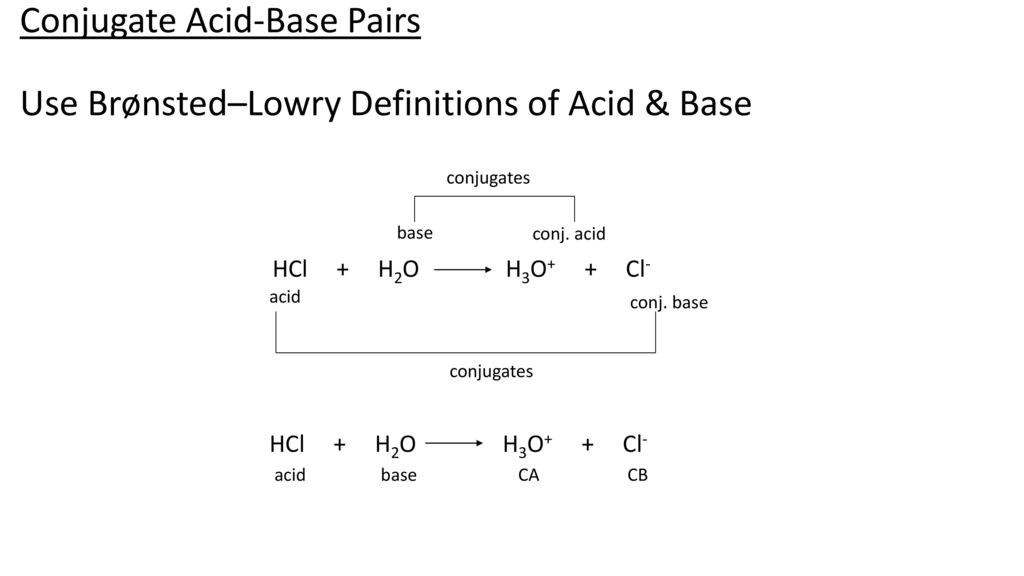
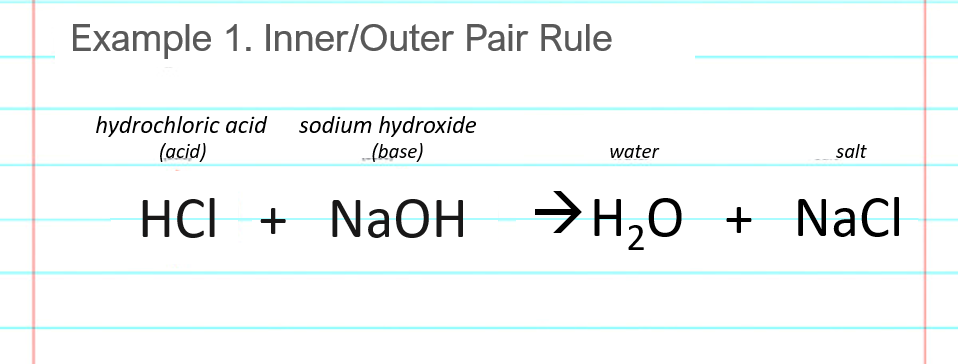
Conjugate Acid-Base Pairs. Acid Dissociation Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 280 HCl Conjugate base Acid Conjugate pair + - ppt download