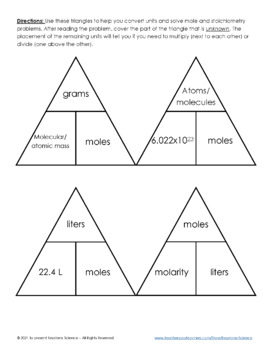
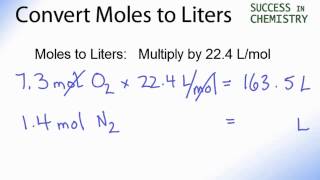SOLVED: What is the total number of moles of solute contained in 0.50 liters of 3.0M HCl? How many liters of a 0.2M NaOH solution would contain 0.4 moles of NaOH? How

SOLVED:Use molar volume to calculate each of the following at STP: a. the volume, in liters, occupied by 2.50 moles of N2 gas b. the number of moles of CO2 in 4.00


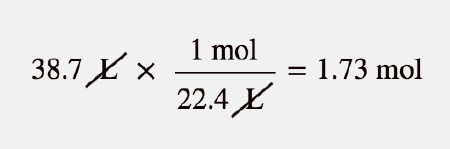




:max_bytes(150000):strip_icc()/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)