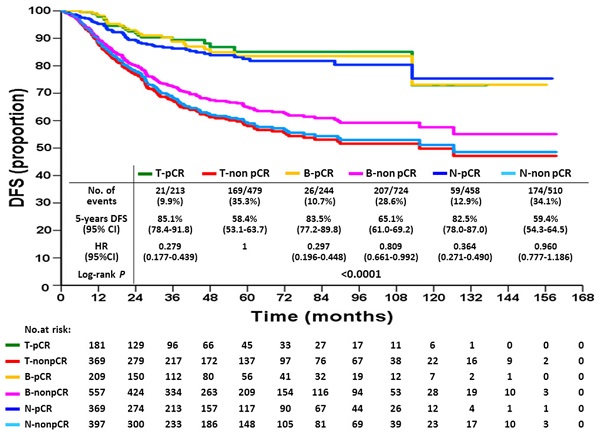
Discrepancy of Breast and Axillary Pathologic Complete Response and Outcomes in Different Subtypes of Node-positive Breast Cancer after Neoadjuvant Chemotherapy

Neo Sphere (Six-Fours-les-Plages) | Avis, Emails, Dirigeants, Chiffres d'affaires, Bilans | 799545389
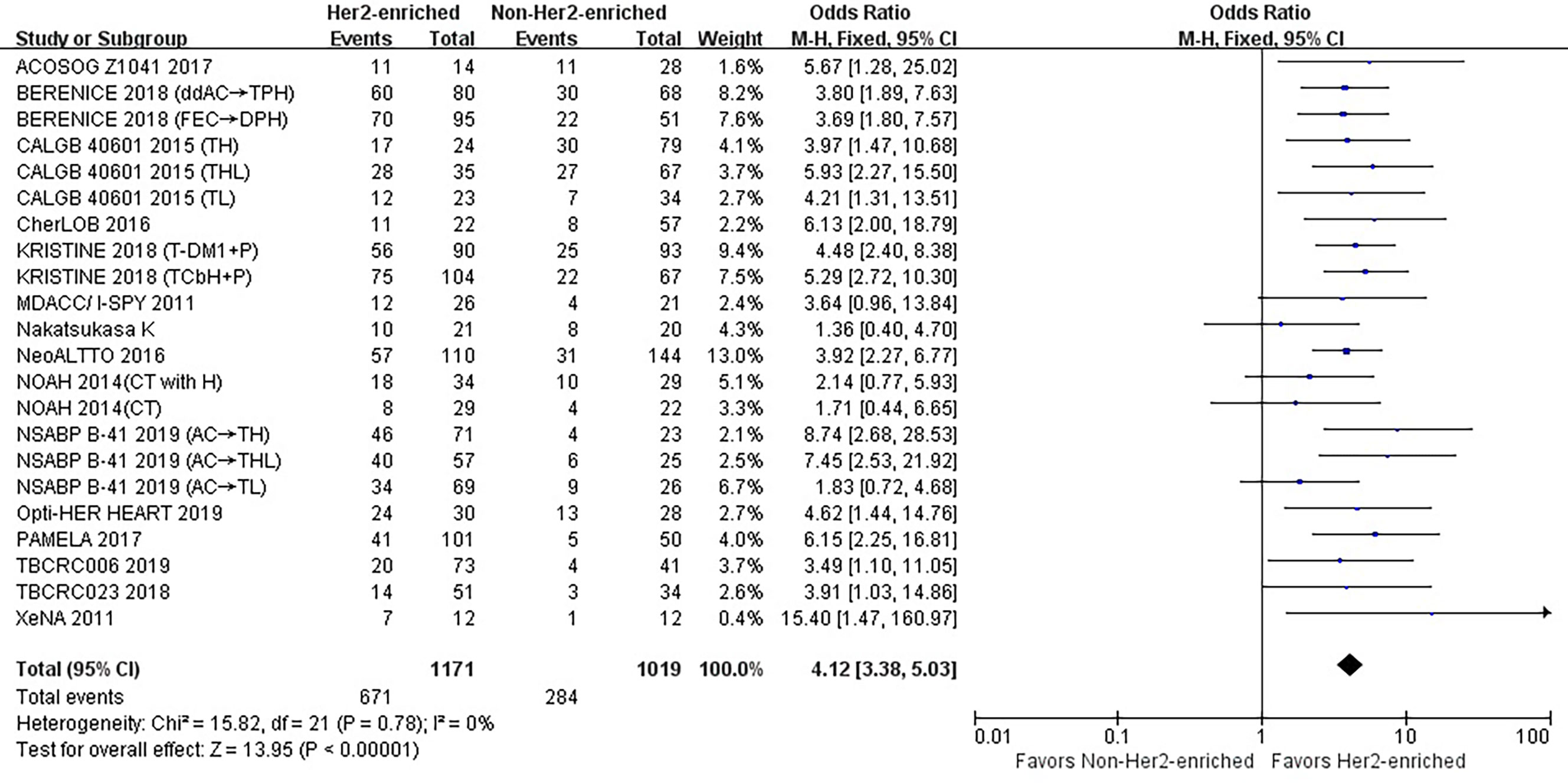
Frontiers | Meta-Analysis of HER2-Enriched Subtype Predicting the Pathological Complete Response Within HER2-Positive Breast Cancer in Patients Who Received Neoadjuvant Treatment

6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (PERSEPHONE): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial - The Lancet

Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer ( NeoSphere): a randomised multicentre, open-label, phase 2 trial - The Lancet Oncology

Incidence and management of diarrhea in patients with HER2-positive breast cancer treated with pertuzumab - ScienceDirect

Immune modulation of pathologic complete response after neoadjuvant HER2-directed therapies in the NeoSphere trial - ScienceDirect
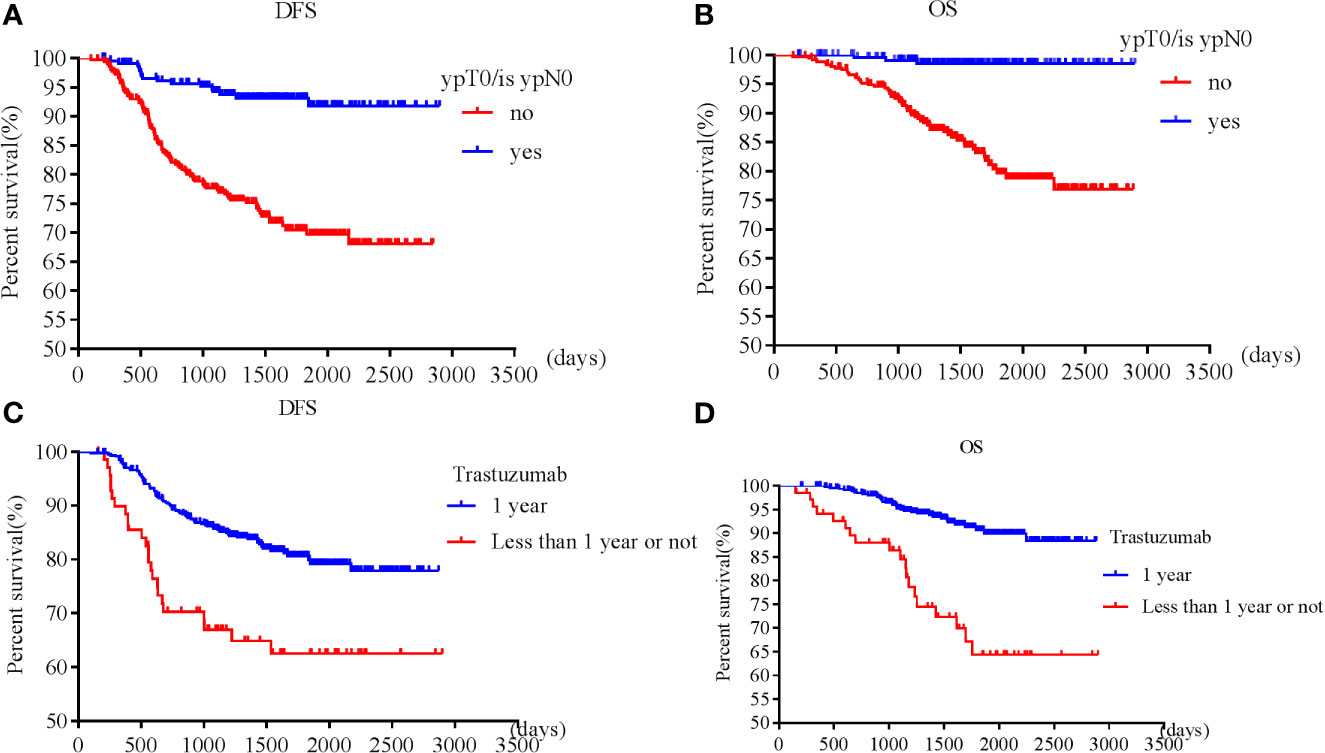
Frontiers | Clinical and Genetic Predictive Models for the Prediction of Pathological Complete Response to Optimize the Effectiveness for Trastuzumab Based Chemotherapy

Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer ( NeoSphere) a randomised multicentre, open-label, phase 2 trial – OncoPrescribe

Neo Sphere - Formation professionnelle, 60 av Joseph Raynaud, 83140 Six Fours les Plages (France) - Adresse, Horaire