
Typical examples for strong Lewis base organocatalysts NHC (A) and TBD... | Download Scientific Diagram
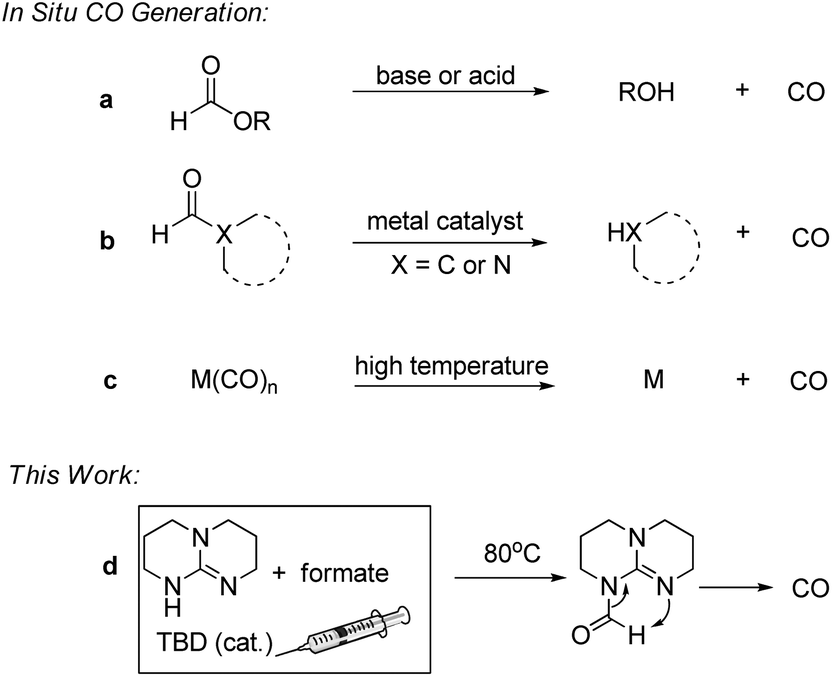
Formates plus triazabicyclodecene (TBD): an efficient platform for non-gaseous carbonylation and unexpected hydrogenation - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00028C

Tbd Da Definire in Base Al Concetto Di Attività Di Acronimo Illustrazione di Stock - Illustrazione di termine, discusso: 214662601
BJOC - A chiral analog of the bicyclic guanidine TBD: synthesis, structure and Brønsted base catalysis
![Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/acs.oprd.2c00248/asset/images/large/op2c00248_0008.jpeg)
Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development
![Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/acs.oprd.2c00248/asset/images/medium/op2c00248_0015.gif)
Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development
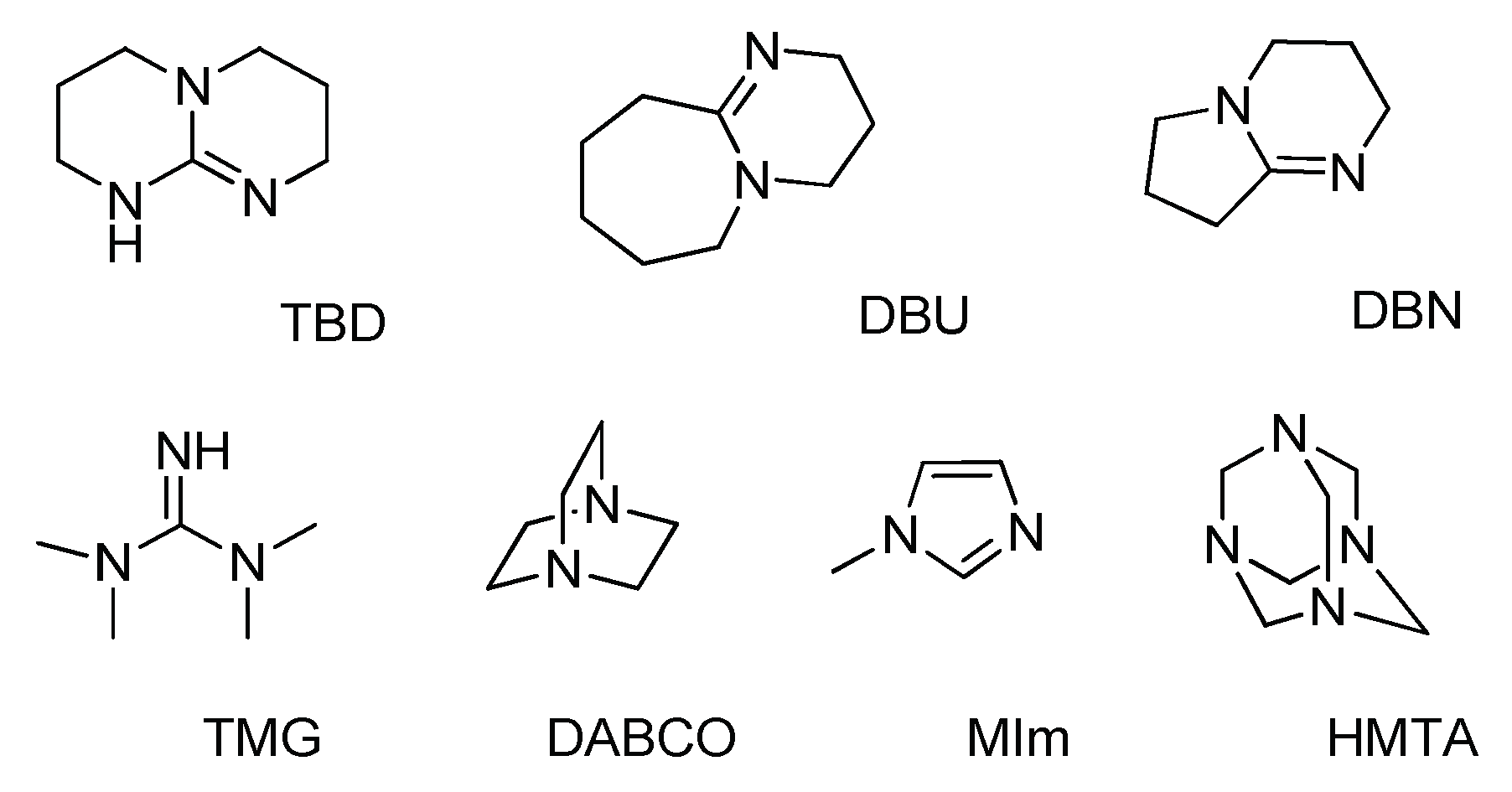
Catalysts | Free Full-Text | Organic Base-Catalyzed C–S Bond Construction from CO2: A New Route for the Synthesis of Benzothiazolones
![Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/acs.oprd.2c00248/asset/images/medium/op2c00248_0001.gif)
Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development
![Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]dec-5-ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry | Organic Process Research & Development](https://pubs.acs.org/cms/10.1021/acs.oprd.2c00248/asset/images/medium/op2c00248_0003.gif)



![1,5,7-Triazabicyclo[4.4.0]dec-5-ene 5807-14-7 | 東京化成工業株式会社 1,5,7-Triazabicyclo[4.4.0]dec-5-ene 5807-14-7 | 東京化成工業株式会社](https://www.tcichemicals.com/medias/T1982.jpg?context=bWFzdGVyfHJvb3R8NDAxNDJ8aW1hZ2UvanBlZ3xoN2IvaDlhLzg5Mjc5MDc5MzgzMzQvVDE5ODIuanBnfDY3NzcwZjkyZjZlNzY3OWU0ZmIxNzNhMzI4NmFhYjBkZjA1YjRhMzQ0NzViNmY1MWE4M2MyZGMxY2IyYzYzNjE)




![7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene (mTBD) Thermal Properties 7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene (mTBD) Thermal Properties](https://thermtest.se/wp-content/uploads/2019/10/backbone-structure-dbu-tbd-mtbd.jpg)


